The expansion of recombinant technology has unleashed the scientific potential of monoclonal antibodies and brought with it improved antibody consistency, sensitivity, control, specificity, and scalability via high-throughput genotype-specialized production.
Recombinant antibodies are monoclonal antibodies produced in vitro from cloned heavy and light chain antibody genes. While the genome sequences are well known for primers or hybridization probe designs, any antibody-producing animal species can have cloned antibody genes to produce recombinant antibodies. Boster Bio featured products can provide the best antibody-related services.
High-yield plasmid DNA vectors transected into hosts such as bacterial, yeast, or mammalian cell lines are used to drive the expression of cloned heavy and light chain genes as single proteins convert to natural IgG in the host-molecules joined together.
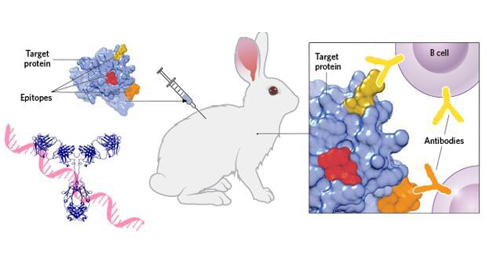
Image Source: Google
Once filtered and isolated, genetically-engineered cell lines can be stored for a long time or used to accelerate the mass production of monoclonal antibodies.
The development of recombinant antibodies involves building an antibody library. The libraries varied depending on the source of the cloned heavy and light chain cDNA sequences. These sources are categorized as: naive, immune, semi-synthetic, or fully synthetic.
The naive and immune libraries represent animal sequences because they are derived from direct cloning of the IgG gene or from unimmunized and immunized animals. Expressing antibodies from immunized animals is universal, established, and healthy because they have undergone in vivo selection and are more likely to exhibit antigen-specific recognition.
However, the use of animal production libraries can have several drawbacks including more labor and time-consuming the first stage of production which relies on the isolation of animal B cells. In contrast, semi-synthetic and fully synthetic libraries derive their IgG sequences from known antibody constructs and the introduction of various sequences into the CDR.
